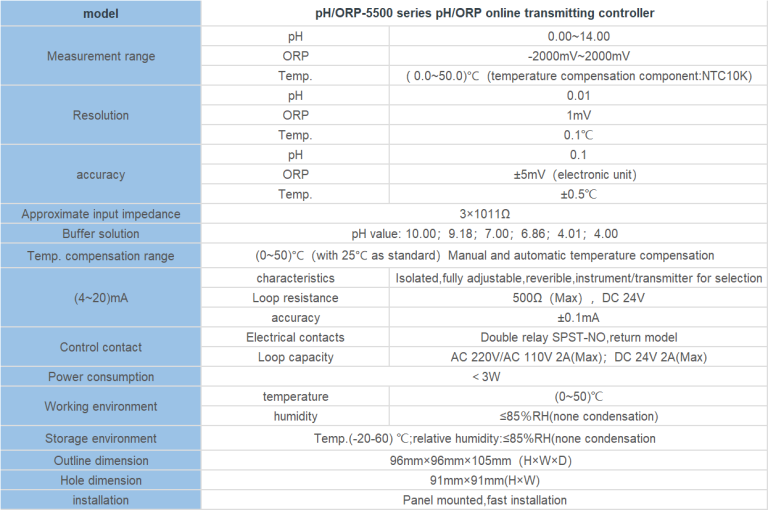
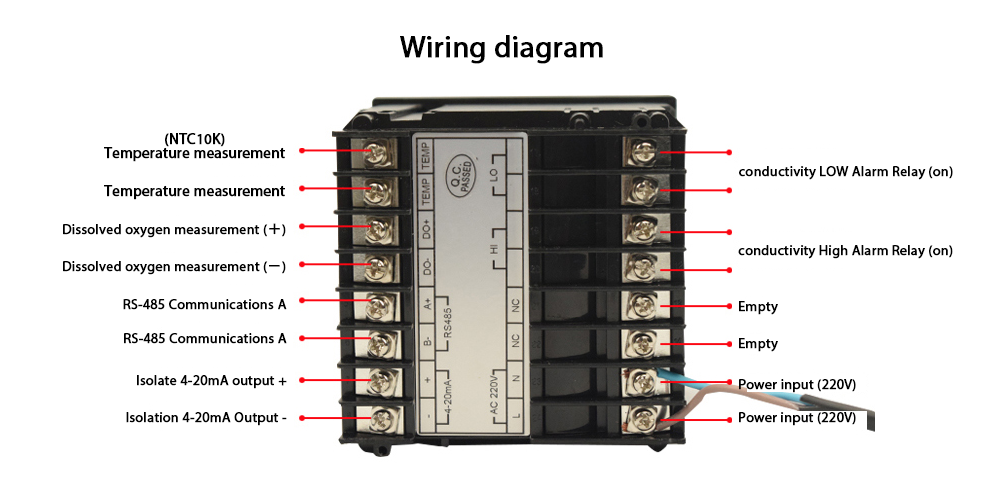
“Temperature’s Impact: Unveiling the Conductivity Connection.”
The Impact of Temperature on Conductivity
The conductivity of a material refers to its ability to conduct electricity. It is a fundamental property that determines how easily electric current can flow through a substance. Conductivity is influenced by various factors, including temperature. In this article, we will explore the impact of temperature on conductivity and understand how it affects the flow of electric current.
When it comes to conductivity, temperature plays a crucial role. In general, as the temperature of a material increases, its conductivity tends to increase as well. This is because higher temperatures provide more energy to the atoms or molecules in the material, allowing them to move more freely. As a result, there is a greater likelihood of electrons being able to move through the material, leading to increased conductivity.
The relationship between temperature and conductivity can be explained by the behavior of electrons within a material. At lower temperatures, electrons have less thermal energy and are more tightly bound to their respective atoms or molecules. This restricts their ability to move freely and hinders the flow of electric current. As the temperature rises, the thermal energy increases, causing the electrons to gain more mobility and enabling them to move more easily through the material.
It is important to note that the impact of temperature on conductivity varies depending on the type of material. In metals, for example, the relationship between temperature and conductivity is relatively straightforward. As the temperature increases, the increased thermal energy causes more electrons to be available for conduction, resulting in higher conductivity. This is why metals are generally good conductors of electricity.
In contrast, the relationship between temperature and conductivity in non-metallic materials, such as semiconductors and insulators, is more complex. In these materials, the behavior of electrons is influenced by the presence of energy bands. At absolute zero temperature, semiconductors have a completely filled valence band and an empty conduction band, resulting in minimal conductivity. However, as the temperature increases, some electrons gain enough energy to move from the valence band to the conduction band, allowing for increased conductivity.
Insulators, on the other hand, have a large energy gap between the valence and conduction bands, making it difficult for electrons to transition from one band to the other. As a result, even at higher temperatures, insulators generally exhibit low conductivity.
It is worth mentioning that while temperature generally has a positive effect on conductivity, there are exceptions to this rule. In certain materials, such as superconductors, the relationship between temperature and conductivity is inverted. Superconductors are characterized by zero electrical resistance at very low temperatures, known as the critical temperature. As the temperature increases beyond this critical point, the conductivity of superconductors rapidly decreases.
In conclusion, temperature has a significant impact on conductivity. In most materials, an increase in temperature leads to an increase in conductivity due to the greater mobility of electrons. However, the relationship between temperature and conductivity can vary depending on the type of material. Metals generally exhibit higher conductivity at higher temperatures, while semiconductors and insulators have more complex behavior. Understanding the influence of temperature on conductivity is crucial for various applications, ranging from electrical engineering to materials science.